Research
Experience-dependent plasticity (e.g. Hebbian plasticity), can be a double-edged sword. It is recognized as the basis for learning and memory, allowing the neural circuit to incorporate new information and adapt to new environments. Due to its self-reinforcing nature, however, Hebbian plasticity can also make the neural circuit susceptible to perturbations and bring instability into the system. Other forms of plasticity (grouped under the general name homeostatic plasticity) work as counteracting mechanisms to keep Hebbian plasticity in check. A long-standing effort in neuroscience aims to understand the cellular and molecular mechanisms underlying different forms of plasticity and how they interact with each other on various temporal and spatial scales in the nervous system. Our long-term goal is to pinpoint cellular and molecular mechanisms that mediate the interaction between experience-dependent plasticity, which shapes and refines the neural circuit, and homeostatic plasticity, which maintains the stability of the circuit.
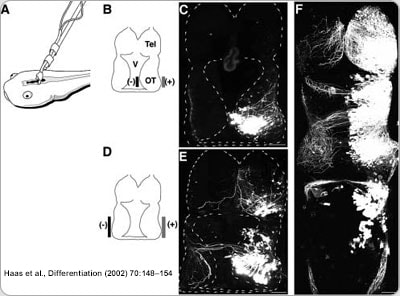
The brain structure we focus on is called the optic tectum (see the picture above), which is the equivalent of the superior colliculus in the mammalian brain. As in the superior colliculus, the optic tectum receives direct inputs from multiple sensory modalities, and is in charge of coordination of sensory-motor integration. One major sensory input processed by the optic tectum is visual information. Retinal ganglion cell axons start innervating optic tectum at the same time when animals start free swimming. This means that the nascent visual circuit needs to be fully functional while at the same time still being actively refined by input visual experience, as all developing sensory circuits are. By manipulating the visual experience the animal receives, or interfering with molecular genetic manipulations, we can facilitate or stall the maturation of the visual circuit, and interrogate how plasticity and stability of the circuit interact from molecular to cellular to circuit level.
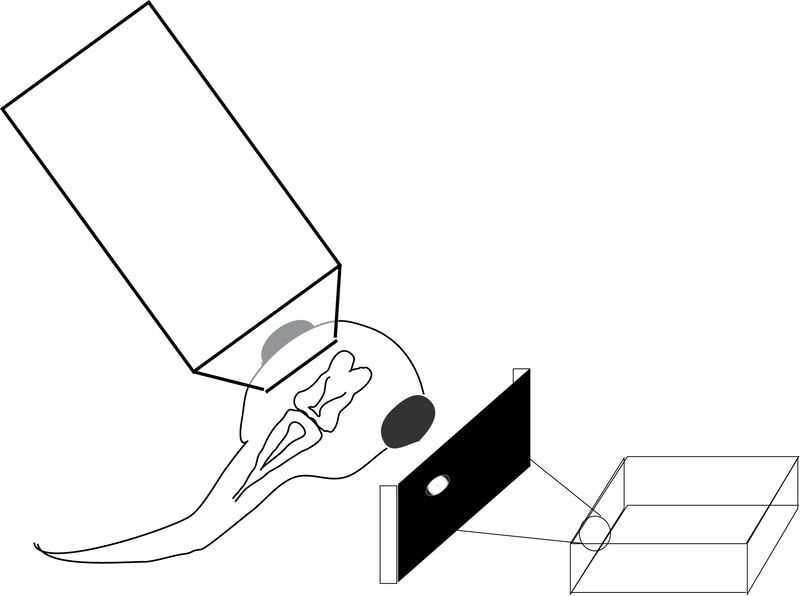
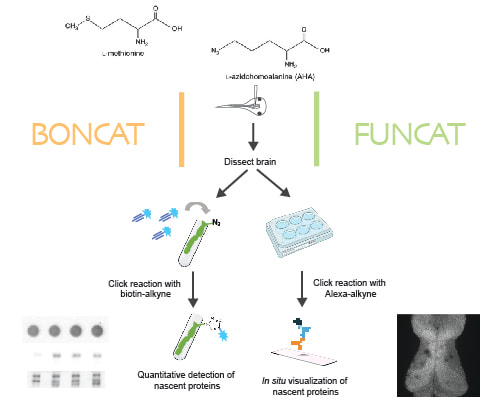
In our lab, we use multidisciplinary tools, from molecular genetics, microscopy, and biochemistry, to time-lapse structural and functional in vivo imaging, as well as behavioral test in live animals, to ask our questions. Click on the above pictures to learn more about each technique.
Activity-dependent proteostasis of nascent proteome
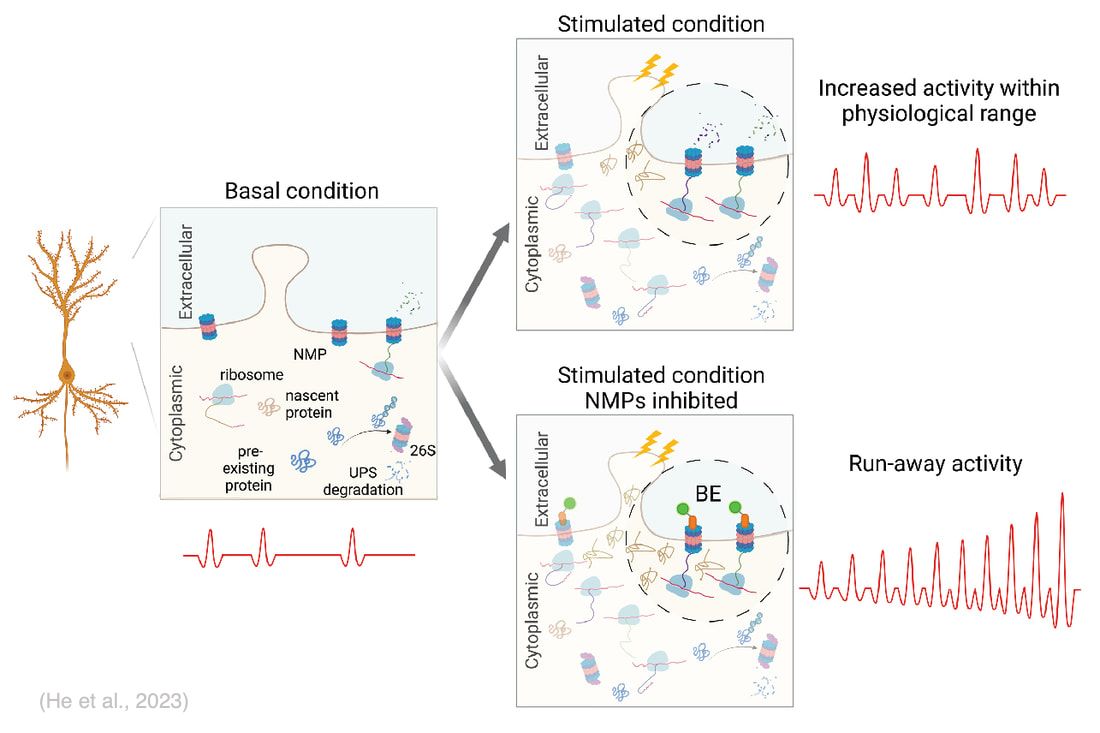
A signature of Hebbian-type plasticity mechanisms is activity-driven de novo synthesis of plasticity-related proteins. A new study from our lab suggests rapid degradation of activity-induced nascent proteins by a recently discovered neuronal membrane proteasome (NMP) may be required for the proper expression of experience-dependent plasticity. NMPs are 20S neuronal-membrane associated proteasomes first discovered in mouse hippocampal neurons (Ramachandran and Margolis, 2017). In neuronal cultures, they were shown to exclusively degrade activity-induced nascent proteins in response to increased neuronal activity (Ramachandran et al., 2018). We have recently established an animal model in Xenopus laevis tadpoles to investigate NMP activity and function in vivo (He et al., 2023). Our data show that NMPs are conserved in the vertebrate nervous system and preferentially degrade nascent proteins in vivo. Inhibiting NMP proteolytic activity in live tadpole brain induced a rapid run-away of neuronal activity. Intriguingly, NMP inhibition ablated learning-induced behavioral improvement in tadpoles in a visuomotor learning paradigm, but did not affect the basal behavior in the absence of training, suggesting that NMP function may be specifically required in the experience-dependent plasticity process. We are currently using well-established plasticity paradigms to investigate the cellular and molecular mechanisms underlying NMP functions in response to plasticity-inducing sensory stimulations.
Development of functional inhibitory neuronal subtypes
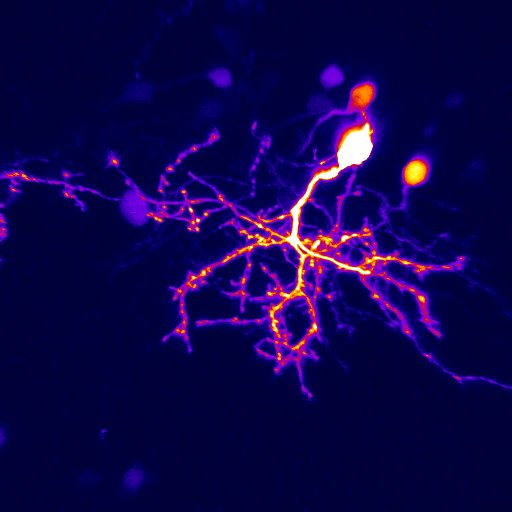
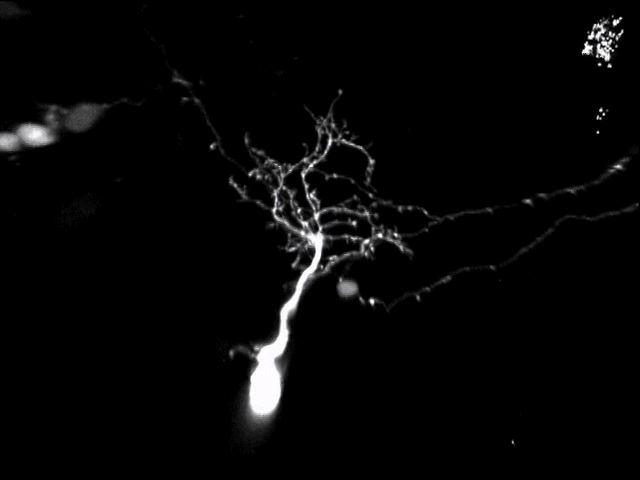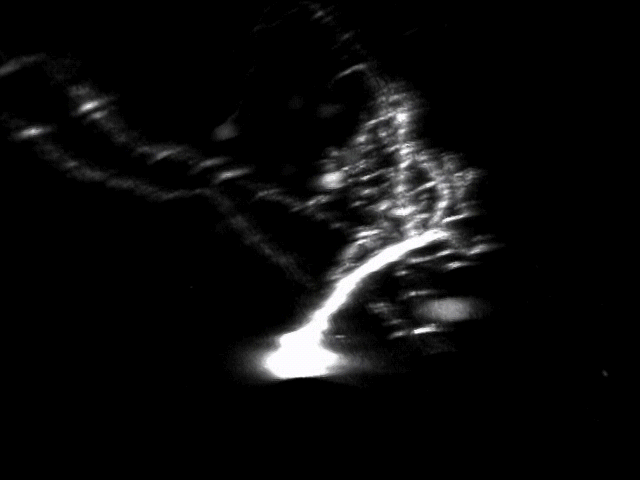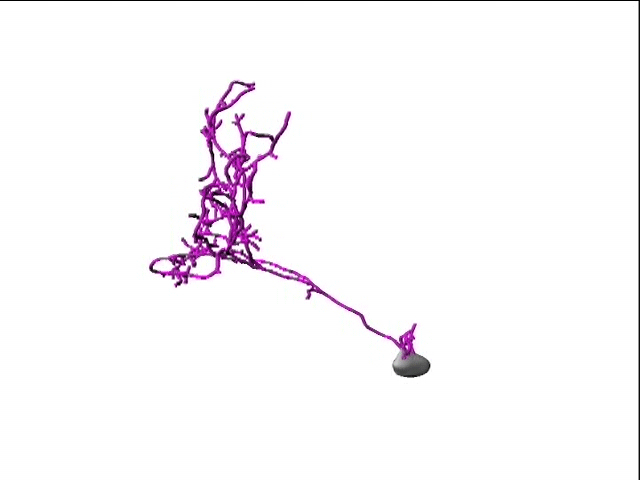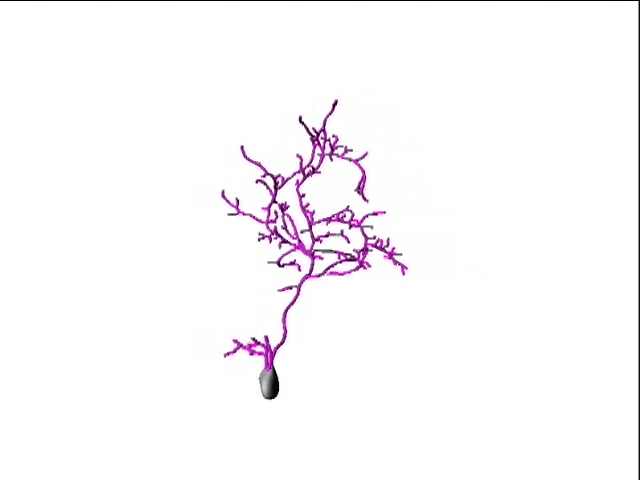
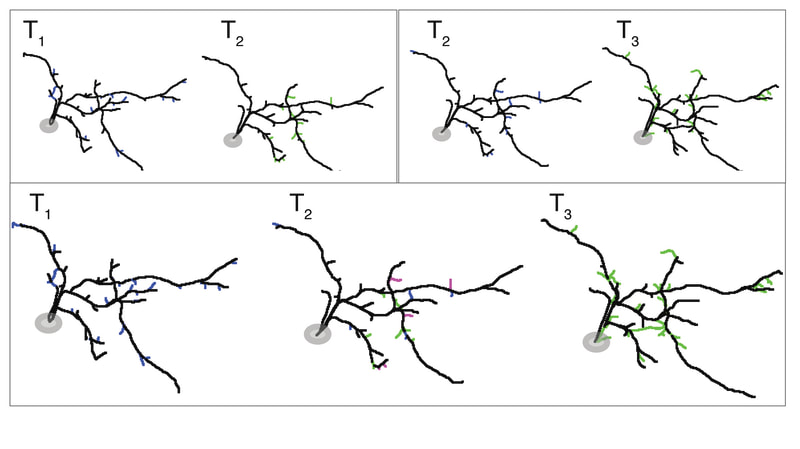
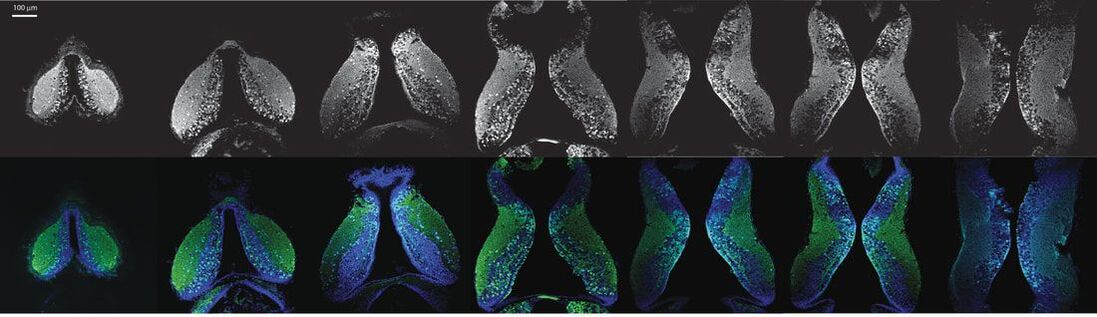
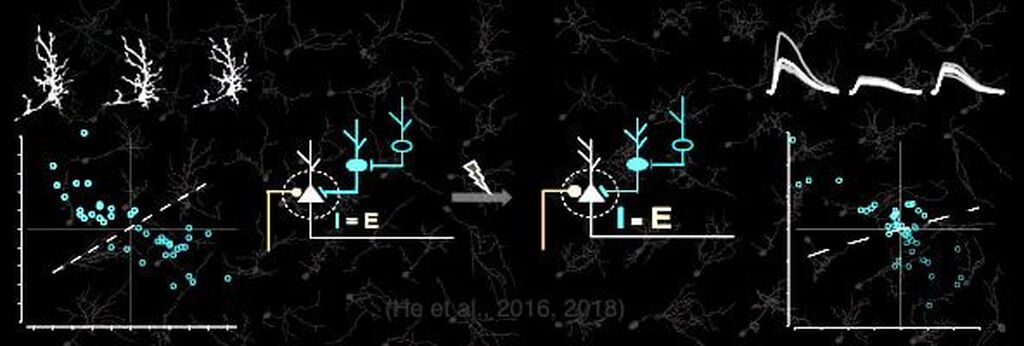
A second active research area in the lab concerns the activity-dependent plasticity of inhibitory neurons during early development and its involvement in the etiology of neurodevelopmental disorders. GABAergic inhibitory neurons comprise about 30% of tectal neuronal population, shown here by GABA immunostaining in serial vibratome sections of the optic tectum (Green: GABA; Blue: Dapi; stage 47). At this developmental stage, tectal inhibitory neurons appear rather homogeneous in their dendritic morphology and electrophysiological properties. However, in response to different visual experiences, the inhibitory neuronal population displays distinct functional heterogeneity: two subgroups of inhibitory neurons with exactly opposite plasticity profiles can be revealed by both structural plasticity of the dendritic arbors and functional plasticity of visually-evoked Ca++ responses, as measured by in vivo time-lapse imaging of individual neurons.
What are these inhibitory neuronal subtypes? Are they intrinsically defined? Would their plasticity profile be affected by prior experience? How are excitatory and inhibitory synaptic inputs coordinated in response to sensory experience during the early developmental stage? … Understanding the role of experience in the formation and maturation of inhibitory neuronal circuits will also provide important insights to devising better therapeutic strategies for neurodevelopmental disorders, as failure to maintain the excitation-inhibition balance has been proposed as a converging etiological mechanism for many neurological diseases.